For the past several weeks I've been running a quiet experiment. Every Sunday at 6 PM Eastern, an AI...


For the past several weeks I've been running a quiet experiment. Every Sunday at 6 PM Eastern, an AI...
.png)
When to Use Each (and When to Combine Them) AI tools are now standard in regulatory affairs, but the...
.png)
How India is reshaping its regulatory approach to AI-enabled medical devices ahead of 2026
.png)
Rethinking UK Market Access: CE Mark Recognition May Be Here to Stay The United Kingdom is once agai...
Predicting a baby’s arrival has always been part science, part estimation. Even with ultrasound imag...
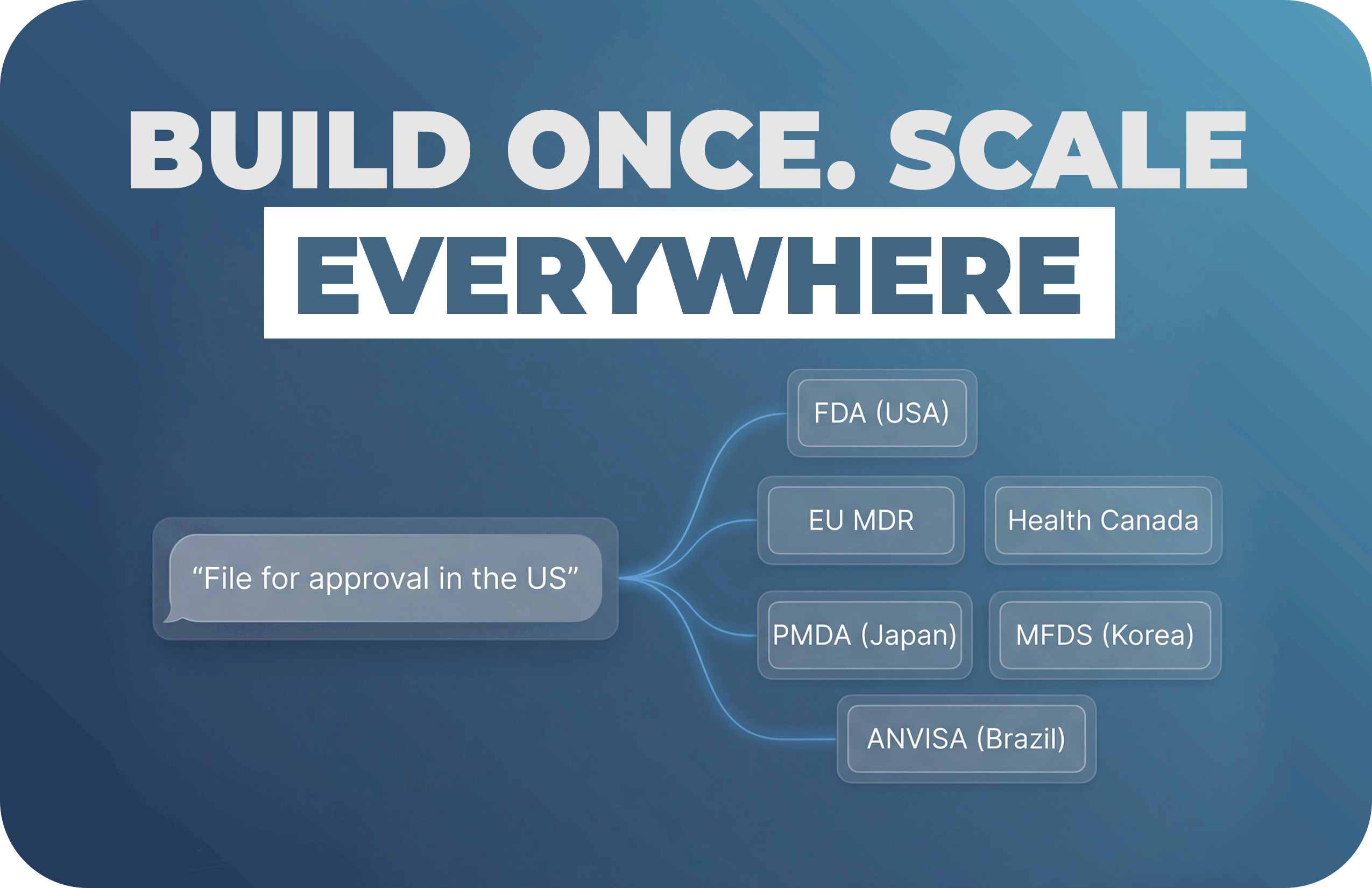
A straightforward plan for teams that want real progress, not another “AI experiment.”

How a Small RA Team Should Measure AI Impact, Choose the Right Level of Adoption, and Navigate Trans...



Case Study: Regulatory Affairs in MedTech